Batteries: Long History, Powerful Future
We tend to think of batteries as a common everyday item. Nevertheless, the concept of batteries - and their changing chemistries - has a fascinating history
By Dr. Benjamin Wilson (Aalto University, Finland)
You are probably reading this piece from a smartphone, tablet or laptop that features some of the most advanced components like a touch screen based on OLEDs or quantum dots, GHz speed CPU processors and multi-megapixel miniature camera, to name but a few. But what about the battery that is powering all this state-of-the-art machinery? Generally, most of us don’t consider the battery technology deployed in our devices except – perhaps – when running out of energy at a critical moment, for instance, when sharing an essential (?) social media update. And even then, it is usually to curse science, which is yet to invent the “everlasting” battery!
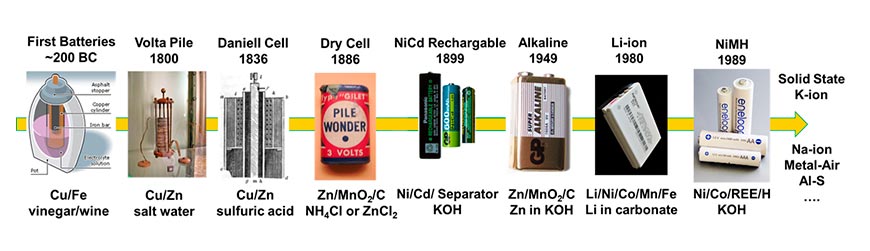
Nevertheless, in parallel with many other technologies in widespread use today, even something as relatively straightforward as the powerpack in your multimedia devices is a cutting-edge technology that has evolved over one or two decades. Although it is true that currently used batteries chemistries – like Li-ion – are the product of over 50 years of continuous development, surprisingly, simple batteries have been discovered to have already been in use by the Parthians somewhere between 190 BCE to 224 CE. Furthermore, there is also circumstantial archaeological evidence in the form of Sumerian silver electroplated vases, found in what is now Southern Iraq, which suggests the first batteries could have been invented as early as 2500 BCE.
So, what were these early batteries like? How do they differ from what we are using today? Essentially, these Antiquarian power sources comprised of an iron rod wrapped in copper, immersed in an acidic electrolyte solution, like wine vinegar, and sealed in a clay pot. Modern testing of replica battery cells demonstrated that such a set-up produces an appreciable output of between 1.5 to 2 V, certainly enough to enable the decorative electroplating of metals found by archaeologists. A similar approach still finds its use today in schools across the globe in the form of the perennial teachers’ favourite: The Lemon (or Potato) Battery that uses two different metal electrodes and a fruit/vegetable of choice as a source of the conductive electrolyte.

After such an impressive start to the idea of batteries, it may come as somewhat of a surprise that it took nearly another 2000 years before the next breakthrough occurred, driven by the industrial revolution and the need for rapid, reliable communication via the telegraph. The three pioneers in this era were Italian, and their names are still synonymous with batteries and electricity even today: Luigi and Lucia Galvani (Galvanostat) and Alessandro Volta (Volts).
The Galvani spouses are most famous for their work with frogs. It was they who, in 1786, accidentally discovered that a combination of static electricity and ions within the nerves allowed a previously dissected frog’s legs to move by “Animal Electricity”! Although this phenomenon was of huge interest to the fledgling medical profession, Volta is the one who correctly identified that the source of the electricity came from two dissimilar metals getting in contact via a moist substance rather than the frog’s leg. He used this knowledge to create the first modern battery in 1800: the Volta Pile, formed of alternating silver and zinc plates separated by fabric or paper soaked in a salt solution.
Since then, there has been comparatively rapid progress in battery technology and complexity. Including the creation of the Daniell Cell in 1820 by British researcher John Daniell, which featured two distinct metal-electrolyte compartments - based on copper-copper sulfate and zinc-sulfuric acid - that allowed for a longer-term current flow. Other advances during this period were primarily related to the use of different chemistries. For example, the Leclanché carbon-zinc cell (1866), the first to incorporate a paste (ammonium chloride) as the electrolyte to produce the first “dry” cell, and the Faure lead acid battery, the basis of which are still used in automotive applications.
Of course, no discussion of such history can be complete without a mention of Thomas Edison, who at the turn of the twentieth century developed one of the first alkaline-based batteries using iron and nickel oxide combined with potassium hydroxide. The first rechargeable batteries based on nickel and cadmium as a material combination were also independently found by Berg and Junger in Sweden around the same time. From then, the Leclanché cell came to dominate the market due to its stability and lifetime. And although the modern alkaline-manganese chemistry was developed by Eveready Batteries in 1949, it still took until the 1960s for these higher-current batteries with increased levels of energy storage to surpass Leclanché’s technology, almost 100 years after its invention.
How about the Nobel Prize-Winning Li-ion batteries that are going to help power the greener transport revolution? These are among the “newer” types of chemistries created by John B. Goodenough, M. Stanley Whittingham, and Akira Yoshino – amongst others – during the 1970s and 1980s. Interestingly and despite the name, the amount of Li is relatively small as the main components of such batteries can generally include cobalt, nickel, graphite, iron, manganese, phosphate and others, depending on the requirements of the final application. Around 1989, Philips developed Nickel Metal Hydride (NiMH) batteries with even more complex chemistries, featuring nickel, cobalt and Rare Earth Elements (REEs) like lanthanum, cerium and yttrium.

What about the future? With the increased awareness of resource scarcity, the origins of some battery materials and the need for more sustainable lifestyles, current research is returning to simpler chemistries based on readily available elements, such as sodium and potassium, as replacements for lithium. Alternative approaches like solid-state, metal-air and Al-S batteries are also under consideration.
Thanks to funding from the European Union’s Horizon 2020 Research and Innovation programme, the HELIOS project integrates innovative circular designs, materials and technologies to improve the charging capabilities, overall performance and cost-effectiveness of electric battery packs for electric vehicles. The HELIOS project believes that a new concept of a standardised, modular and scalable battery pack, which will pave the way to a more sustainable - close-to-zero emissions - urban transportation, is possible.
As for an “everlasting” battery, although this remains a potentially [un]realistic objective, it may have already been created! The Oxford Electric Bell Experiment, which started in 1840 and consists of two brass bells, each with a dry pile battery and metal sphere clapper, is still ringing today. Unfortunately, the nature of the battery chemistry remains a mystery, as its exact content got lost in the mists of time.